Abstract
Background: Patients should be informed beforehand of the risk factors for exocrine pancreatic insufficiency (ExoPI) after pancreatic surgery; however, there are no clear identified risk factors for this condition. This study aimed to identify the preoperative, perioperative and postoperative risk factors for ExoPI after pancreatic surgery.
Methods: We conducted a systematic search of PubMed, Scopus, SAGE, CINAHL Plus and Taylor & Francis from inception to Mar. 7, 2021, for full-text articles that included patients who had undergone pancreatic surgery. The primary outcome was the number of ExoPI events and any risk factors evaluated. We used the Newcastle–Ottawa Scale to assess study quality.
Results: Twenty studies involving 4131 patients (2312 [52.3%] male, mean age 60.12 [standard deviation 14.07] yr) were included. Of the 4131 patients, 1651 (40.0%) had postoperative ExoPI. Among the 11 factors evaluated, the significant risk factors were preoperative main pancreatic duct (MPD) diameter greater than 3 mm (odds ratio [OR] 4.50, 95% confidence interval [CI] 1.06–19.05), pancreaticoduodenectomy (PD) as the surgical treatment procedure (OR 3.31, 95% CI 1.92–5.68), pancreaticogastrostomy (PG) as the anastomotic procedure (OR 3.13, 95% CI 1.83–5.35), hard pancreatic texture (OR 2.93, 95% CI 1.99–4.32) and adjuvant chemotherapy (OR 2.50, 95% CI 1.54–4.04). Gender, history of diabetes mellitus or endocrine pancreatic insufficiency (EndoPI), underlying diseases, de novo diabetes or EndoPI, pylorus-preserving PD and postoperative pancreatic fistula were not risk factors for ExoPI after pancreatic surgery.
Conclusion: Preoperative MPD diameter greater than 3 mm, PD, PG reconstruction, hard pancreatic texture and adjuvant chemotherapy were risk factors for the development of ExoPI after pancreatic surgery. The findings should provide useful information for patients to reduce postoperative dissatisfaction and improve quality of life.
Exocrine pancreatic insufficiency (ExoPI) is defined as a condition of insufficient production of pancreatic substances in the presence of partial or complete deficit of acinar and ductal functions. The usual clinical presentations of ExoPI include steatorrhea, abdominal pain, flatulence and bloating.1,2 The condition is classified as mild, moderate or severe, based on the fecal elastase-1 level.3
Surgical methods such as the Whipple procedure (pancreaticoduodenectomy [PD]) and distal (or left) pancreatectomy (DP or LP) are considered safe for treating benign or malignant pancreatic diseases.4 The occurrence of ExoPI after surgery is high, with a prevalence of about 19%–80% with DP and 56%–98% with the Whipple procedure.1,5
Several risk factors for postsurgery ExoPI considered to be related to the surgical procedure itself have been identified.4,5 However, there are no clearly standardized and established risk factors in these studies.4 Moreover, the quality of life of patients who undergo pancreatic resection is affected soon after surgery. Although their quality of life improves from time to time, Schniewind and colleagues6 found that they did not experience a better quality of life than before surgery. Thus, in this study, we performed a meta-analysis with the aim of identifying the preoperative, perioperative and postoperative risk factors for ExoPI after pancreatic surgery.
Methods
Search strategy
This systematic review and meta-analysis were conducted in accordance with the PRISMA statement.7 We searched PubMed, Scopus, SAGE, the CINAHL Plus database in EBSCOhost, and Taylor & Francis for articles that included patients who had undergone pancreatic surgery. Computerized data searching of the relevant studies was conducted independently by all authors from inception to Mar. 7, 2021. Keywords were constructed based on Medical Subject Headings terms and other additional terms as follows: ((“exocrine pancreatic insufficiency”) OR (“pancreatic exocrine insufficiency”) OR (“EPI”) OR (“PEI”) OR (“ExoPI”) OR (“pancreatic exocrine dysfunction”) OR (“pancreatic function”)) AND ((“pancreatic surgery”) OR (“pancreatic resection”) OR (“pancreatectomy”) OR (“pancreaticoduodenectomy”) OR (“pancreatoduodenectomy”)) AND ((“risk factor”) OR (“risk factors”) OR (“predictive factors”)). The search was limited to human participants; there was no language restriction. The protocol of this study was registered in the PROSPERO international prospective register of systematic reviews (CRD42021252617).
Eligibility criteria, data extraction and quality assessment
Studies were included if they were observational studies, the study population consisted of patients undergoing any pancreatic surgical procedure, and the measurable outcome was the number of ExoPI events and any possible risk factors evaluated. The exclusion criteria were irrelevant title or abstract, irretrievable full-text article, article in a language other than English and incorrect study design (review article, case report, case series, letter to the editor or conference abstract). We extracted the author and year of publication, study location, study design, ExoPI diagnostic method or definition, study population, surgical procedure, sample size, age of patients, risk factors evaluated and number of ExoPI events. We assessed the quality of the selected studies using the Newcastle–Ottawa Scale. Any disagreements were resolved through group discussion involving all authors. For cohort studies, those with a Newcastle–Ottawa Scale score of 7–9 were considered to be of good quality, those with a score of 4–6, of moderate quality, and those with a score of 3 or less, of poor quality. The quality of cross-sectional studies with a Newcastle–Ottawa Scale score of 9–10 was considered “very good,” a score of 7–8 “good,” a score of 5–6 “satisfactory,” and a score of 4 or less “unsatisfactory.”8
Statistical analysis
We performed all statistical analyses using RevMan 5.4 (The Cochrane Collaboration). Meta-analysis of ExoPI events was performed in 3 phases: 1) preoperative factors (gender, history of diabetes mellitus or endocrine pancreatic insufficiency [EndoPI], underlying diseases and preoperative main pancreatic duct [MPD] diameter), 2) perioperative factors (surgical procedures, anastomotic reconstruction procedures and pancreatic texture) and 3) postoperative factors (de novo diabetes mellitus or EndoPI, postoperative pancreatic fistula and adjuvant chemotherapy).
We assessed heterogeneity among studies with the χ2 test (Cochran Q statistic) and quantified it with the I2 statistic. An I2 value less than 25% was considered as low heterogeneity, of 25%–75% as moderate heterogeneity, and of greater than 75% as high heterogeneity. If the I2 value was greater than 50%, we used a random-effect model for the meta-analysis; otherwise, a fixed-effect model was used. We transformed median age (and interquartile range) from the included studies into mean and standard deviation (SD) using a standardized online calculator (http://www.math.hkbu.edu.hk/~tongt/papers/median2mean.html) or manually using a standardized mathematical formula if the study provided only mean, minimum and maximum.9,10 A p value < 0.05 was considered statistically significant. We assessed publication bias using a funnel plot when there were more than 10 studies included in the meta-analysis. We performed a sensitivity analysis to assess the influence of each individual study on the pooled subgroup and the overall results using the leave-one-out approach.
Results
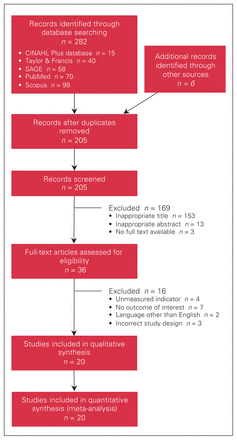
The initial literature search yielded 282 studies obtained from the databases searched and 6 studies obtained from other sources (Figure 1). Of the 288 studies, 205 were screened by title and abstract after duplicates were removed. Thirty-six full-text articles were reviewed based on the eligibility criteria, of which 16 were excluded owing to unmeasured indicators (n = 4), no identified outcome of interest (n = 7), language other than English (n = 2) or incorrect study method (n = 3). Twenty studies were thus included for both qualitative and quantitative synthesis.
Flow diagram showing study selection.
A summary of the included studies is provided in Table 1. The 20 studies involved 4417 patients (2312 [52.3%] male, mean age 60.12 [SD 14.07] yr) who underwent pancreatic surgery. Of the 4417, 4131 (93.5%) were assessed for ExoPI; 1651 ExoPI events (40.0%) were identified. Of the included studies, 18 were retrospective or prospective cohort studies, and 2 were cross-sectional studies.2,26 The studies were conducted in various locations such as the United States, Asia and Europe. The diagnostic methods or definitions of ExoPI and therapeutic operative procedures varied across studies. All included studies were considered as being of good or very good quality (Table 2).
Basic characteristics of included studies
Assessment of quality of included studies with the Newcastle–Ottawa Scale8
Risk factors for exocrine pancreatic insufficiency
Five factors were found to be risk factors for development of ExoPI after pancreatic surgery: preoperative MPD diameter, surgical procedure, anastomotic or reconstruction procedure, pancreatic texture and adjuvant chemotherapy.
Preoperative main pancreatic duct diameter
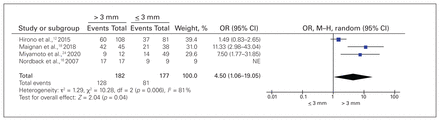
Four studies with a total of 359 patients who had undergone therapeutic pancreatic surgery were included in the analysis of preoperative MPD diameter. Evidence of high heterogeneity was detected (I2 = 81%); therefore, we used a random-effect model for the analysis. The pooled odds ratio (OR) indicated that patients who had a dilated preoperative MPD greater than 3 mm had about 4.5 times the risk for postoperative ExoPI compared to patients without MPD dilation (MPD ≤ 3 mm) (OR 4.50, 95% confidence interval [CI] 1.06–19.05) (Figure 2).
Odds ratio (OR) of preoperative main pancreatic duct diameter greater than 3 mm versus 3 mm or less in patients with exocrine pancreatic insufficiency events after pancreatic surgery (359 patients). CI = confidence interval; M–H = Mantel–Haenszel; NE = not estimable.
Surgical procedure
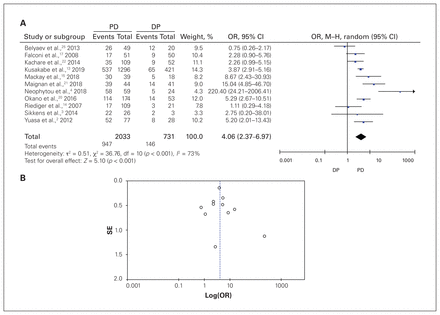
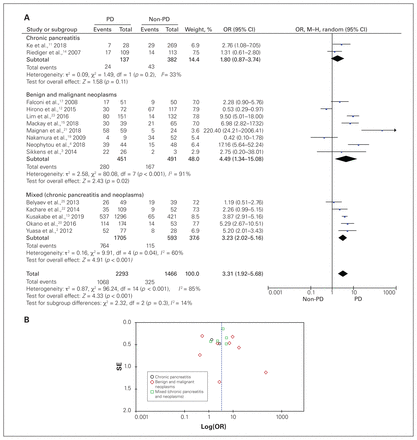
The pooled OR of PD versus DP involving 2764 patients in 11 studies showed that, compared to patients who had DP, those who had therapeutic PD had 4 times the risk of developing ExoPI postoperatively (OR 4.06, 95% CI 2.37–6.97), with high heterogeneity (I2 = 73%) (Figure 3A). A subgroup meta-analysis of PD versus non-PD involving 3759 patients in 15 studies showed that PD was a risk factor for the development of postoperative ExoPI in the neoplasm subgroups (OR 4.49, 95% CI 1.34–15.08) and mixed-patient groups (OR 3.23, 95% CI 2.02–5.16), as well as in the overall pooled analysis (OR 3.31, 95% CI 1.92–5.68), but not in the chronic pancreatitis groups (OR 1.80, 95% CI 0.87–3.74) (Figure 4A). The random-effect model was applied since there was high overall heterogeneity (I2 = 85%). The test for subgroup differences suggested that there was no statistically significant sub-group effect (p = 0.3). The funnel plot showed asymmetric distribution, indicating potential publication bias (Figures 3B and 4B).
Odds ratio (OR) of therapeutic surgical procedures in patients with exocrine pancreatic insufficiency events after pancreatic surgery. (A) Pancreaticoduodenectomy (PD) v. distal pancreatectomy (DP); (B) funnel plot of PD v. DP. CI = confidence interval; M–H = Mantel–Haenszel; SE = standard error.
Odds ratio (OR) of therapeutic surgical procedures in patients with exocrine pancreatic insufficiency events after pancreatic surgery. (A) Disease-based subgroup analysis of pancreaticoduodenectomy (PD) v. non-PD, (B) funnel plot of PD v. non-PD. CI = confidence interval; M–H = Mantel–Haenszel; SE = standard error.
Anastomotic or reconstruction procedure
A total of 334 patients from 4 studies were evaluated in the analysis of anastomotic or reconstruction procedure. There was no evidence of heterogeneity across studies (I2 = 0%); therefore, a fixed-effect model was applied for the analysis. The pooled analysis showed that, compared to pancreaticojejunostomy, pancreaticogastrostomy (PG) as the anastomotic reconstruction procedure was a significant risk factor for postoperative development of ExoPI (OR 3.13, 95% CI 1.83–5.35) (Figure 5).
Odds ratio (OR) of anastomotic or reconstruction procedure in patients with exocrine pancreatic insufficiency events after pancreatic surgery (334 patients). CI = confidence interval; M–H = Mantel–Haenszel.
Pancreatic texture
Five studies with a total of 564 patients who had undergone therapeutic pancreatic surgery were included in the analysis of pancreatic texture. Evidence of low heterogeneity was detected (I2 = 0%); therefore, a fixed-effect model was used for the analysis. The pooled OR (2.93, 95% CI 1.99–4.32) indicated that patients who had hard pancreatic texture were at about 2–3 times higher risk for postoperative ExoPI compared to patients with soft pancreatic texture (Figure 6).
Odds ratio (OR) of hard versus soft pancreatic texture in patients with exocrine pancreatic insufficiency events after pancreatic surgery (564 patients). CI = confidence interval; M–H = Mantel–Haenszel; NE = not estimable.
Adjuvant chemotherapy
A total of 774 patients from 5 studies were included in the analysis of adjuvant chemotherapy. Evidence of high heterogeneity was detected (I2 = 51%); therefore, a random-effect model was applied for the analysis. The pooled OR indicated that patients who received postoperative adjuvant chemotherapy had about 2.5 times the risk for developing postoperative ExoPI compared to patients who did not receive adjuvant chemotherapy (OR 2.50, 95% CI 1.54–4.04) (Figure 7).
Odds ratio (OR) of postoperative adjuvant chemotherapy in patients with exocrine pancreatic insufficiency events after pancreatic surgery (774 patients). CI = confidence interval; M–H = Mantel–Haenszel.
Factors that were not risk factors for exocrine pancreatic insufficiency
Six factors were found not to be risk factors for development of ExoPI after pancreatic surgery: gender, history of diabetes or EndoPI, underlying diseases, de novo diabetes or EndoPI, pylorus-preserving PD and postoperative pancreatic fistula.
A total of 1621 patients from 11 studies were included in the analysis of gender. We used a fixed-effect model for the analysis since there was no evidence of heterogeneity in the data (I2 = 0%). The pooled analysis showed that male gender was not a risk factor for development of ExoPI after pancreatic surgery (OR 1.09, 95% CI 0.88–1.36) (Appendix 1, Figure S1A, available at www.canjsurg.ca/lookup/doi/10.1503/cjs.010621/tab-related-content). The funnel plot showed an asymmetric distribution, suggesting potential publication bias (Appendix 1, Figure S1B).
A total of 1001 patients in 6 studies were included in the analysis of history of diabetes or EndoPI. Heterogeneity across the studies was detected (I2 = 53%); thus, a random-effect model was used for the analysis. The pooled analysis showed that a history of diabetes or EndoPI was not a risk factor for ExoPI after pancreatic surgery (OR 1.56, 95% CI 0.90–2.69) (Appendix 1, Figure S2).
A total of 595 patients from 4 studies and 625 patients from 5 studies were included in the analysis of underlying diseases as potential risk factors: neoplasms versus chronic pancreatitis, and malignant versus benign neoplasms, respectively. Both analyses showed high heterogeneity (I2 = 90% and 68%, respectively); therefore, random-effect models were applied. The pooled analysis showed that, compared to chronic pancreatitis, the presence of neoplasms was not a risk factor for the development of ExoPI after pancreatic surgery (OR 0.92, 95% CI 0.18–4.86) (Appendix 1, Figure S3A). Similar results were found for malignant neoplasms compared to benign neoplasms (OR 1.06, 95% CI 0.50–2.22) (Appendix 1, Figure S3B).
A total of 975 patients from 5 studies were included in the analysis of de novo diabetes or EndoPI. A fixed-effect model was used for the analysis since heterogeneity in the data was not detected (I2 = 0%). The pooled analysis showed that having de novo diabetes or EndoPI postoperatively was not a risk factor for ExoPI after pancreatic surgery (OR 1.16, 95% CI 0.85–1.57) (Appendix 1, Figure S4).
The pooled analysis of 309 patients in 3 studies showed that, compared to standard PD, pylorus-preserving PD was not a risk factor for postoperative development of ExoPI (OR 1.59, 95% CI 0.97–2.63), with low heterogeneity (I2 = 33%) (Appendix 1, Fig. S5).
Five studies involving 491 patients were included in the analysis of postoperative pancreatic fistula. A fixed-effect model was used for the analysis since low heterogeneity in the data was detected (I2 = 28%). The pooled analysis showed that having postoperative pancreatic fistula was not a risk factor for the development of ExoPI after pancreatic surgery (OR 0.77, 95% CI 0.42–1.42) (Appendix 1, Figure S6).
Sensitivity analysis
The results of the sensitivity analysis suggested that the statistical significance of pooled subgroups and overall point estimates in several meta-analyses were not affected by any single study. However, when we excluded several studies (that by Maignan and colleagues21 or Miyamoto and colleagues24 in the meta-analysis of preoperative MPD diameter, that of Belyaev and colleagues25 in the meta-analysis of neoplasms v. chronic pancreatitis, that of Ke and colleagues11 in the meta-analysis of pylorus-preserving PD v. PD, and that of Hirono and colleagues12 in the meta-analysis of anastomotic or reconstruction procedure), the pooled ORs were changed, from significant to nonsignificant, or from nonsignificant to significant. The latter sensitivity analysis indicated that the statistical significance of pooled ORs was affected by several studies.
Discussion
In this meta-analysis, preoperative MPD diameter greater than 3 mm, PD, PG reconstruction, hard pancreatic texture and adjuvant chemotherapy were found to be risk factors for the development of ExoPI after pancreatic surgery.
In more than half of the patients with pancreatic cancer, the mass was located in the head of the pancreas. In addition, other neoplasms growing in the lumen of the duct had the potential to occlude the duct and develop fibrosis, thus resulting in exocrine insufficiency.5 With a similar outcome, chronic pancreatitis cases also contribute to a high prevalence of de novo exocrine insufficiency owing to the process of necrosis, apoptosis, inflammation and duct obstruction. Ultimately, these conditions cause the destruction of secretory parenchyma and a reduction in exocrine or endocrine production.27 These possible occurrences may predict long-term pancreatic obstruction after surgery, especially in early pancreatic duct obstruction.28 Our findings indicate that patients with chronic pancreatitis were at slightly higher risk for developing de novo ExoPI than patients with neoplasms. This may be due to the possibility of fibrosis development in the former.29
We found that a dilation process greater than 3 mm adds to the risk of developing ExoPI. Diffuse dilation of the MPD is often related to atrophy and fibrosis of the glandular parenchyma.30 Dilated MPD with a diameter greater than 3 mm is associated with a hard-textured pancreas, which reflects a higher fibrosis ratio; this may play a role in the development of exocrine insufficiency after surgery.31
Pancreaticoduodenectomy involves removal of the pancreatic head, common bile duct, gallbladder, duodenum and proximal jejunum, which may anatomically disrupt the flow of pancreatic enzymes, causing an insufficiency.32 Nakamura and colleagues19 also reported atrophy of the pancreatic remnants after surgical intervention, which suggests that PD indirectly reduced the function of the pancreas. This may be mediated by the reduced serum gastrin level because of the duodenal resection.
Hard pancreatic texture may be attributed to fibrosis. Pancreatic fibrosis is also known to cause parenchymal atrophy and duct dilation, thus causing exocrine insufficiency.30 Nakamura and colleagues19 also suggested that pre-existing pancreatic obstruction and duct dilation cause the pancreas to become harder and contribute to ExoPI.
Patients with PG are more vulnerable to ExoPI because of the process of enzyme degradation by the gastric contents, as lipase is easily destroyed by the acid.33 Gastric juice may also cause chronic inflammation of the anastomotic site tissue in patients who undergo PG.34 This condition induces pancreatic tissue atrophy or stenosis, and further contributes to the development of ExoPI.34
During the last decades, much progress has been made in adjuvant treatments for pancreatic cancer. As a result, the median postsurgical survival has more than doubled.35 Interestingly, we found that adjuvant chemotherapy was a risk factor for postsurgery ExoPI. Although the evidence explaining the relation between the two is still unclear, we found a hypothesis suggesting that it is probably related to burnout of the pancreas owing to the underlying disease.23 Alternatively, adjuvant chemotherapy may induce a fibrotic reaction in the pancreas and further cause a decrease in the pancreatic remnant function.23
We recognize that most of the risk factors identified in this study cannot be greatly modified. Nevertheless, our findings may help clinicians deliver clearer and more comprehensive explanations to patients before surgery regarding which factors may present risk for the development of ExoPI. Early detection of and regular examination for MPD dilation and hard pancreatic texture are encouraged so that they can be closely monitored. In addition, chronic pancreatitis is associated with progressive development of fibrosis, which ultimately results in a harder pancreatic texture.29,30 Hence, we suggest that surgical intervention for patients with chronic pancreatitis should be performed as early as possible to prevent further risk of ExoPI. Furthermore, since we also found adjuvant chemotherapy to be a risk factor, we suggest that its use should be carefully considered by weighing the benefits and risks for the individual patient. If the benefits outweigh the risks, adjuvant chemotherapy may be used.
Limitations
The pooled number of patients included in each analysis was relatively sufficient, supported by the high quality of the included studies and a wide range of geographic areas. Limitations include the fact that the underlying pancreatic disease and the type of neoplasms were varied in and among studies. Subgroup analyses based on the disease could not be conducted for most of the risk factors. Second, the definitions of ExoPI were heterogeneous between the studies, and the condition was not clearly defined in 1 study.24 Third, publication bias was observed in our study. This may have been due to the fact that studies are more likely to be published if they present significant findings. Fourth, only English-language articles were included, and pertinent research published in other languages may not have been identified. This may be another cause of the observed publication bias, since studies with nonsignificant findings are also more likely to be published in a local journal in a language other than English.36
Conclusion
We found risk factors for postoperative ExoPI at each phase of pancreatic surgery. A wide diameter (> 3 mm) of the main pancreatic duct was a risk factor in the preoperative phase. Pancreaticoduodenectomy compared to DP, PG compared to pancreaticojejunostomy reconstruction, and hard pancreatic texture were perioperative risk factors, and adjuvant chemotherapy was a risk factor in the postoperative phase. We hope that the findings provide useful information for patients to reduce postoperative dissatisfaction and improve quality of life.
Footnotes
Competing interests: None declared.
Contributors: V. Budipramana, A. Witarto and B. Witarto designed the study. All authors acquired the data, which V. Budipramana, A. Witarto and B. Witarto analyzed. All authors wrote the manuscript, which V. Budipramana, A. Witarto, B. Witarto, S. Pramudito and L. Ratri critically revised. All authors gave final approval of the article to be published.
- Accepted February 9, 2022.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/