Abstract
Background: The incidence of colorectal cancer (CRC) is increasing among young adults. We sought to report on patient and disease characteristics, treatment practice patterns and outcomes in this population.
Methods: We conducted a retrospective cohort study using administrative health data from the Alberta Cancer Registry (2004–2015), including demographic and tumour characteristics, and treatment received. Outcome measures included overall and cancer-specific deaths. We used Cox regression and Kaplan–Meier curves to assess for factors associated with survival.
Results: We included 18 070 patients with CRC (n = 1583 [8.8%] < 50 yr, n = 16 487 [91.2 %] ≥ 50 yr). Younger patients were more likely to present with locally advanced disease (21.0% v. 18.0%, p < 0.0001), stage III (16.4 % v. 14.6%, p < 0.0001) or metastatic (16.7% v. 13.8%, p < 0.0001) involvement. Younger patients were more likely to receive surgery (87.2% v. 80.9%, p < 0.0001), chemotherapy (59.6% v. 34.1%, p < 0.0001) or radiation therapy (49.5% v. 37.2%, p < 0.001). At 5 years, overall and cancer-specific survival was better among younger patients than older patients (30.6% v. 51.5% overall deaths, 27.5% v. 38.4% cancer-specific deaths, p < 0.0001).
Conclusion: Despite higher stage and higher grade disease, young patients with CRC had more favourable oncologic outcomes than stage-matched older patients, which may be related to younger patients receiving more aggressive treatment. Further investigation should focus on optimal treatment patterns for young patients with CRC.
Colorectal cancer (CRC) is the third most commonly diagnosed malignant disease in both the United States and Canada.1,2 Although epidemiologic reports have shown a decline in CRC in those older than 50 years, new evidence has shown an increase in the incidence of CRC among young adults in nearly a dozen high-income countries.2,3 In Canada, the incidence of CRC in adults younger than 50 years has increased by a mean annual percentage change of 4.45% for males since 2010 and by 3.47% for females since 2006.4,5 Several factors have been postulated to account for the increase in CRC among young people, such as the concomitant rise in the incidence of obesity; however, no specific risk factors have been identified to selectively target screening for younger individuals.4 About 10% of cases of CRC are now diagnosed in people younger than 50 years.3–6
Young patients with CRC may have a biologically distinct disease and unique treatment considerations, compared with older patients with CRC. Recent literature suggests that young patients with CRC are more likely to present with locally advanced disease, nodal involvement and metastases at initial diagnosis.7–11 Despite this, some studies suggest that young patients still go on to have better outcomes than stage-matched older patients.12 In addition, previous studies have suggested that young patients with CRC may be overtreated.13 Thus, an ongoing debate remains with respect to whether younger patients are being appropriately treated.14
We sought to report on patient and disease characteristics, treatment practice patterns and oncologic outcomes for young patients who received diagnoses of CRC in Alberta, Canada, over a 10-year period using provincial administrative health data.
Methods
We used deidentified, population-based, administrative data from the Alberta Cancer Registry between the period of 2004 and 2015. The registry records and maintains data on all new cancer cases and cancer-related deaths in Alberta, and is regularly audited to ensure accuracy, completion and timeliness of data entry. Reporting of all cancer cases occurring in the province is mandated by the Regional Health Authorities Act of Alberta. We performed deterministic linkage to Alberta Vital Statistics (a division of Statistics Canada).
Patient population
We identified all patients with CRC in the province of Alberta using codes from the Canadian version of the International Classification of Disease, 10th Revision, Canada (ICD-10-CA), namely C18–20 (malignant neoplasms of the colon through rectum) and C26.0 (malignant neoplasm of the intestinal tract, part unspecified). We excluded patients with hereditary polyposis syndrome (ICD-10-CA code D12.6) or inflammatory bowel disease (ICD-10-CA code K51.90).
Study variables
We collected patient characteristics, including sex, age, year of diagnosis, Charlson Comorbidity Index, average neighbourhood income and education. We extracted disease characteristics — including stage (tumour, lymph node, metastasis [TNM] and American Joint Committee on Cancer [AJCC]), tumour grade and tumour location (colon, sigmoid or rectum) — and treatment practice patterns, including health care facility type (community hospital v. academic centre) where treatment was received and treatment received (surgery, chemotherapy, radiation therapy). We collected clinical outcomes, including average follow-up duration, death and cancer-specific death.
Statistical analysis
We expressed descriptive statistics of continuous variables as means and standard deviations. We used 2-tailed Student t tests to make comparisons between 2 groups and analysis of variance with appropriate post hoc testing to compare differences between 3 or more groups. We considered p values less than 0.05 statistically significant. We expressed categorical variables as frequencies, and compared groups using the χ2 test. We used the Kaplan–Meier method to estimate overall survival. We performed multivariate Cox regression analysis to report on factors that affected overall survival. We adjusted the survival analysis and the Kaplan–Meier curves by sex, year of diagnosis, Charlson Comorbidity Index, cardiovascular disease diagnosis, type of health care facility, cancer size, cancer stage, cancer grade, receipt of chemotherapy, receipt of radiation therapy, receipt of surgery, tumour location, education and income. We generated the adjusted survival curves by plotting survival function estimates on the event times for each stratum.15 We performed statistical analysis using Stata version 14 software (StataCorp).
Ethics approval
Ethics approval of this study was granted by the Health Research Ethics Board of Alberta Cancer Committee (HREBA.CC-19–0123).
Results
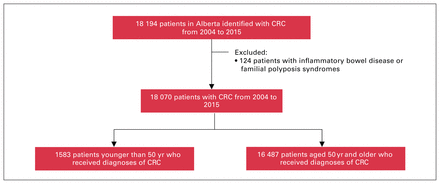
A total of 18 194 patients received diagnoses of CRC in Alberta during the 2004–2015 period (Figure 1). Of these patients, 124 were identified as having either a polyposis syndrome or inflammatory bowel disease and were excluded from the analysis, leaving 18 070 patients included in the cohort (Figure 1). Of those included, 1583 (8.8%) patients were younger than 50 years and 16 487 (91.2%) were 50 years of age or older (Table 1). There were no differences in proportion by sex between the younger and older cohort. In the younger cohort, the average age at diagnosis was 41.9 (standard deviation [SD] 7.0) years. Most young patients were otherwise healthy, with 63.5% having a Charlson Comorbidity Index of 0 or 1, compared with 47.6% (p < 0.0001) for the older cohort.
Study flowchart. CRC = colorectal cancer.
Characteristics of patients who received diagnoses of colorectal cancer in Alberta from 2004 to 2015
Disease characteristics
Younger patients were more likely to present with locally advanced, T4 disease (21.0% v. 18.0%, p < 0.0001) than older patients. Younger patients were also more likely to present with stage III (16.4% v. 14.6%, p < 0.0001) and metastatic (16.7% v. 13.8%, p < 0.0001) disease at diagnosis (Table 1). Patients younger than 50 years had a significantly higher proportion of rectal tumours than older patients (35.6% v. 26.4%, p < 0.0001) (Table 1). Complete staging data were unavailable in 33.2% of younger patients and 35.6% of older patients, so we performed sensitivity analyses, with similar results (Appendix 1, Tables 1–3, available at canjsurg.ca/lookup/doi/10.1503/cjs.019721/tab-related-content).
Treatment practice patterns
Both younger and older patients with CRC received treatment at academic and community hospitals in a near equal distribution, with 53.1% of younger patients receiving treatment in an academic centre compared with 51.2% of older patients (p = 0.1363) (Table 2). Overall, younger patients were more likely to undergo surgery (87.2% v. 80.9%, p < 0.0001) and receive chemotherapy (59.6% v. 34.1%, p < 0.0001) (Table 2). This was particularly true among younger patients with stage III or stage IV cancer (Table 2). When looking specifically at patients with rectal cancer, younger patients were more likely to receive radiation therapy (64.0% v. 50.9%, p < 0.0001) than the older cohort (Table 2). Young patients with stage II cancer were more likely to receive chemotherapy (49.0% v. 19.7%, p < 0.0001).
Treatment practice patterns of patients who received diagnoses of colorectal cancer in Alberta from 2004 to 2015
Outcomes
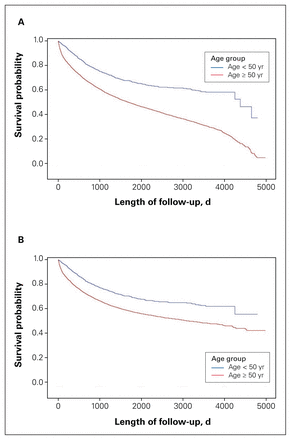
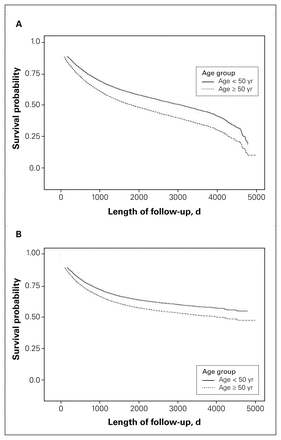
The mean length of follow-up for younger patients was 3.9 (SD 3.2) years. During the follow-up period, the overall mortality rate was 30.6% (27.5% cancer-specific rate) for patients younger than 50 years, compared with 51.5% (38.4% cancer-specific rate) for patients aged 50 years and older ( p < 0 .0001 ) (Table 1). Unadjusted Kaplan–Meier regression curves showed a median overall survival of 4379 days (12.0 yr) for younger patients and 1665 days (4.6 yr) for older patients (p < 0.0001) (Figure 2). Adjusted multivariable Kaplan–Meier regression curves continued to show better overall and cancer-specific survival among younger patients (p < 0.001) (Figure 3).
Unadjusted Kaplan–Meier curves for (A) overall and (B) cancer-specific survival.
Multivariable Kaplan–Meier curves for (A) overall and (B) cancer-specific survival, adjusted for sex, Charlson Comorbidity Index, year of diagnosis, mean neighbourhood income and education level, tumour location, stage, grade, receipt of surgery, receipt of chemotherapy, receipt of radiation therapy and institution where treatment was delivered.
Multivariable regression showed that male sex, year of diagnosis, a Charlson Comorbidity Index of 2, tumour grade (2 or 3), not receiving surgery and undergoing treatment in a community centre negatively affected overall survival significantly for younger patients (Table 3).
Multivariate Cox regression analysis of variables affecting overall survival for patients younger than 50 years with colorectal cancer
We performed a sensitivity analysis that excluded patients with missing staging information. The results of the analysis were not substantially different, with the exception of the effect of treatment centre, which was no longer significant (Appendix 1, Table 3).
Discussion
In response to the increased incidence rates of early-onset CRC, it is important that we further understand patient and disease characteristics, treatment practice patterns and oncologic outcomes as they pertain to this relatively poorly understood disease process.
Our population-based analysis complements much of the contemporary literature in this area. In keeping with other studies, we found that 9% of all CRC diagnoses in Alberta over a 10-year period occurred in patients younger than 50 years. Currently in Canada, these patients are younger than those targeted by screening guidelines for average-risk people.16 We found that younger patients were more likely to present with locally advanced cancer, with nodal and metastatic involvement at initial diagnosis. In keeping with other studies, we showed a higher distribution of rectal cancers in the younger cohort, which contributes to discussions regarding screening recommendations for patients younger than 50 years and the potential use of flexible sigmoidoscopy in this population.
In addition, our analysis offered several nuanced findings that warrant further discussion. Younger patients were more likely to receive surgery and chemotherapy than their older counterparts on a stage-by-stage basis. This continues to be an area of discussion, as formal treatment guidelines specific to the treatment of young patients with CRC have yet to be outlined. A recent retrospective analysis of the National Cancer Database by Birkett and colleagues14 showed that young patients with stage II CRC were more likely to receive adjuvant chemotherapy, whether they were of normal or high risk, without a survival benefit. Little guidance exists regarding treatment, and potential overtreatment, of young patients, and this may increase the risk of morbidity, acute toxicity and late adverse effects of chemotherapy in this population. Despite an overall mortality rate of 31% over a mean follow-up period of 3.9 years, our Kaplan–Meier regression did show better overall and cancer-specific survival in younger patients with CRC, compared with older patients. This is consistent with a recent systematic review by Griffiths and colleagues,17 which found that young patients with CRC in North America have improved survival when controlling for cancer stage. It remains unclear if this survival benefit is owing to the ability of young patients to tolerate more aggressive treatment practices or differing cancer biology. Prediction modelling of oncologic outcomes based on specific patient and tumour characteristics could help guide individual treatment for young patients with CRC. Optimally, a prospective cohort study of young patients with CRC should be performed to identify prognostic factors, similar to the Reducing the Burden of Breast Cancer in Young Women (RUBY) study.18
Our multivariate regression analysis showed that male sex, less recent diagnosis (in contrast with patients who received diagnoses more recently), a Charlson Comorbidity Index of 2, tumour grade (2 or 3), not receiving surgery and receiving treatment in a community hospital negatively affected overall survival for young patients. Although some of these variables were expected, outcome disparities related to treatment centre warrant further discussion. There is certainly potential for confounding as young patients with CRC who present to their local community hospital may do so because of emergent complications. In addition, treatment setting was not significant in our sensitivity analyses. However, other data support regional variation in cancer care, both in Alberta and in Canada more broadly.19–22 Patients in community-based sites may have variable access to necessary resources such as cancer centres and imaging, and this may contribute to variable outcomes in this population. In line with the action plan of the Canadian Partnership Against Cancer, further attention should be given to supporting high-quality surgical oncology care, eliminating barriers, establishing pan-Canadian benchmarking strategies and integrating surgical and medical care.22
Limitations
We conducted a retrospective study, with all of the inherent limitations to that study design. The data were limited to routinely collected variables in the Alberta Cancer Registry. Although our use of the registry allowed us to evaluate a large sample size and disease, treatment and outcome patterns, administrative data lack the detail and nuances that are present in pathology, operative and specialist reports. Related to this, we had a substantial amount of missing data with respect to staging. The rate of missing data was consistent over the entire period of study, with no improvement after a particular year, but there certainly could be a nonbiased pattern to the missing data. Our median follow-up time of 10 years is relatively short, which prevents us from reporting on longer-term overall and cancer-specific mortality, which may be relevant to the younger patient. Finally, given that the data are from the census level, there is risk of geographic bias (assuming the overall characteristics of a certain area apply to an individual living in the area). Future work is needed to risk stratify analyses and identify select patients who may benefit from more aggressive treatment measures and centralized referral to tertiary centres.
Conclusion
Younger patients with CRC present with more advanced, higher-grade disease, yet have more favourable oncologic outcomes than their older counterparts. Younger patients were also more likely to be treated aggressively. Future work should not only focus on early identification, but understanding the optimal treatment strategies in this population.
Acknowledgement
The authors acknowledge members of the Alberta Cancer Outcomes Research Network (ACORN) at the University of Calgary for their contributions to this work.
Footnotes
Competing interests: Ameer Farooq is the associate digital editor for CJS; he was not involved in the review or decision to accept this manuscript for publication. He is also codirector of social media for Evidence Based Reviews in Surgery. No other competing interests were declared.
Contributors: A. Farooq and A. MacLean designed the study. W. Cheung acquired the data, which A. Keehn, Y. Xu, S. Kong and M.L. Quan analysed. A. Farooq and A. Keehn wrote the article, which all authors revised critically for important intellectual content. All authors gave final approval of the version to be published and agreed to be accountable for all aspects of the work.
- Accepted June 21, 2022.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/