Abstract
Background Video-assisted thoracic surgery (VATS) can be performed through 1 or more intercostal or subxiphoid ports. The aim of this study was to evaluate whether number and location of ports had an impact on early perioperative outcomes and postoperative pain after anatomical lung resection (ALR).
Methods A search of the departmental electronic database identified all patients who underwent VATS ALR between June 2018 and June 2019. We stratified patients according to the surgical approach: 2-port VATS, 3-port VATS, and subxiphoid VATS. We extracted demographic and clinicopathologic data. We used univariate analysis with unpaired t tests and χ2 tests to compare these variables between the subgroups.
Results We included 201 patients in the analysis. When patients were stratified by surgical approach, there was no difference in terms of age, disease load, length of surgery, postoperative complications, duration of pleural drainage, and length of hospital stay. Postoperative pain and morphine equivalent usage were also comparable between the groups. According to these results, number and location of VATS ports seemingly has no clinical impact on early postoperative outcomes. Limitations of the study include its retrospective nature, small sample size, and short follow-up interval.
Conclusion Our results suggest that incision location and the number of VATS ports is not associated with differences in the incidence of perioperative complications or postoperative pain. Given the limitations described above, further studies with longer follow-up intervals are required to explore the lasting impact of this surgical approach on quality of life.
It was estimated that 30 000 people in Canada would receive a lung cancer diagnosis in 2022.1 Depending on tumour type, stage, and location, surgery may be offered as either the primary modality of treatment or as part of a multimodal approach. Over the last 2 decades, video-assisted thoracic surgery (VATS) has become the standard of care for performing anatomical lung resections (ALR), including lobectomy and segmentectomy.
Since the introduction of VATS as a surgical technique, surgeons have been modifying this approach with a view to reducing operative trauma. With the reduction of the number of ports from 3 to 1, a uniportal lobectomy has become technically possible.2 Proponents of this approach think that a decrease in the number of incisions could lead to a reduction in postoperative pain and lower risks of postoperative complications. Furthermore, considering that chronic neuropathic pain after VATS is well described, efforts to reduce the number of ports is also thought to be beneficial in this regard.3,4
Despite these potential benefits, when different intercostal VATS approaches are compared, data regarding postoperative outcomes have been conflicting. For instance, some authors did not find a significant difference in the postoperative pain scores between uniportal and multiportal VATS.5,6 Conversely, others describe reduced postoperative pain and paresthesia, shorter hospital stays, and less intraoperative blood loss with a uniportal approach.7,8
More recently, the subxiphoid approach has gained in popularity. It is hypothesized that this approach could prevent the development of chronic wound pain by avoiding intercostal incisions and trauma to the intercostal nerves. An additional benefit to this approach is a perceived wider range of motion not limited by the confines of the intercostal space. In canine models, the subxiphoid approach was found to be comparable to transintercostal approaches in terms of mean operating time, intraoperative complication, and postoperative complication.9 Despite the lack of data comparing various surgical approaches, we hypothesize that subxiphoid VATS will be associated with decreased postoperative pain and, similarly, both minor and major complications. The aim of this study was to evaluate the impact of the number and the location of VATS ports on early perioperative outcomes and postoperative pain in patients undergoing an ALR for lung cancer.
Methods
Study setting
A single institution retrospective cohort review was performed at a university-affiliated tertiary referral centre for thoracic surgery based in Montréal, Canada. The study was approved by the McGill University Health Centre Research Ethics Board.
Study design and participants
We identified all patients who underwent a VATS ALR for lung cancer between June 2018 and June 2019. This included patients with either pathological confirmation or a radiologically suspicious lesion. We verified the patients according to the following inclusion criteria: ALR must have been performed, including either a lobectomy or segmentectomy; surgery must have been performed on an elective basis; and patients must have had electronic medical records available for review. Treatment with neoadjuvant chemotherapy or radiotherapy did not preclude inclusion in the study. However, we excluded patients who underwent planed thoracotomy or bilobectomy. In addition, patients who underwent conversion to open surgery were identified and excluded from the analysis.
Data collection
The primary aim of this study was to explore whether there was any difference in perioperative outcomes and postoperative pain scores when stratifying between different surgical approaches. To achieve this, we reviewed patients’ electronic and paper medical records. We reviewed the operation note and noted the type and length of surgery. We calculated the disease stage based on the final pathological report according to the American Joint Committee on Cancer (AJCC) 8th edition of the TNM (tumour, node, and metastasis) Staging System classification. 10 Additionally, we recorded the estimated blood loss. We also reviewed the inpatient notes and calculated the duration of pleural drainage. We extracted complications data from an online database maintained in real time, graded according to the Thoracic Morbidity and Mortality system (see ottawatmm.org). This is based on the Clavien–Dindo classification scheme and has been validated against the American College of Surgery National Surgical Quality Improvement Program (NSQIP) database.11 We noted the incidence of major and minor postoperative complications. We defined a major complication as all complications occurring within 30 days of surgery with a Clavien–Dindo grade of 3 or higher. We recorded the level of postoperative pain in 2 ways. First, we noted the subjective pain score reported by the patients on a visual analogue scale (VAS). The ward’s nursing staff routinely collected this information every 4 hours; 1 referred to no pain and 10 referred to the highest level of pain possible. The mean score for each time of the day (am, pm, and evening) were used in this analysis for the first 3 postoperative days. Second, by reviewing the inpatient drug charts, we used morphine equivalents for the first 3 postoperative days as an objective measurement of pain. We calculated morphine equivalents as a 1.5:1 ratio for morphine and oxycodone and 5:1 for morphine and hydromorphone. We averaged these data per 24-hour period.
Surgical approach
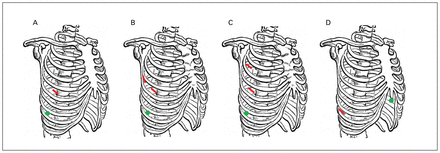
We compared 3 different surgical techniques. The 2-port approach (2p-VATS) involved a 5-mm port in the midaxillary line of the eighth intercostal space, with another 3-cm incision at the level of the fifth intercostal space of the anterior axillary line. The 3-port approach (3p-VATS) involved the same ports as the 2p-VATS, but with an additional 0.5- to 1-cm port in the posterior axillary line, ideally in the fifth intercostal space or in the midaxillary line of the third intercostal space. Because both of these 3p-VATS techniques involved a total of 3 ports, we grouped them together for the analysis. Finally, a subxiphoid approach (Sx-VATS) involved a 3-cm access port in the subxiphoid region with a 5-mm intercostal port, typically inserted in the eighth intercostal space (Figure 1). During the study period, 3 surgeons used the 2p-VATS approach, of whom 1 also performed the Sx-VATS approach. Two additional surgeons used the 3p-VATS approach. A chest tube was inserted at the end of the procedure and removed only once there was no air leak and serous drainage of less than 400 mL per day.
Illustration of the locations of the ports used for each approach on the right side. Locations are shown for the (A) 2-port video-assisted thoracic surgery (VATS) approach, for the (B, C) 3-port VATS approach, and (D) for the subxiphoid VATS approach. The green circles show the 3-cm ports for the camera; red lines show the 0.5- to 1-cm accessory ports.
Postoperative pain management
Routine practice in our centre is that for all patients undergoing VATS procedures, regional analgesia — consisting of multilevel intercostal nerve blocks with 20 mL of 0.5% ropivacaine mixed with 1% epinephrine and 10 mg of dexamethasone — was routinely used. This was injected under thoracoscopic guidance once thoracoscopy had been established. Postoperatively, patients were prescribed a combination of acetaminophen (1 g orally every 6 h), celecoxib (200 mg orally every 24 h), and hydromorphone (2 mg orally every 4 h as required).
Statistical analysis
We performed all statistical analyses in SPSS 27.0 (IBM Corp.). Descriptive statistics used to describe the study population are presented as means or frequency with standard deviation or percentage in parentheses, as appropriate. The percentages for the total number of each approach were based on the overall number of patients. The remaining percentages were based on the total number of patients per category. We performed a univariate analysis of the cohort, stratified by surgical subtype, using independent Student t tests for continuous variables or χ2 for categorical data. A p value < 0.05 was considered statistically significant. We created the figures using Microsoft Excel Version 16.59 (Microsoft).
Results
During the study period, 216 patients underwent a VATS ALR for lung cancer. We excluded 4 patients owing to incomplete data and 11 as they were converted to open. We therefore analyzed 201 patients in total. Of these, 97 patients (48.3%) were treated with a 2p-VATS approach, 87 (43.2%) with a 3p-VATS, and 17 (8.5%) with a Sx-VATS approach. A univariate analysis of the basic demographic and clinical data for the cohort, stratified by surgical approach, is shown in Table 1. The overall mean age was 69 years. We noted no differences in terms of patient age, gender, body mass index, smoking history, and Charlson Comorbidity Index when comparing between the groups. Similarly, we observed no differences with respect to forced expiratory volume in 1 second, nor the diffusing capacity for carbon monoxide between the different surgical approaches. Segmentectomies and lobectomies were performed in 18 and 195 patients, respectively (8.5% and 91.5%). Although there was some variation in the number of procedures performed via each approach, these failed to reach a level of statistical significance. Among the 11 patients who were converted to open, 8 were initially undergoing 2p-VATS, 2 were undergoing 3p-VATS, and 1 was undergoing Sx-VATS. The reasons for conversion are shown in Table 2.
Patient characteristics (N = 201) according to surgical approach
Reason for conversion divided by surgical approach
The operative data are presented in Table 3. The combined mean operating time for the cohort as a whole was 119 minutes, with no difference between the subgroups in terms of operating time or blood loss. The length of chest drainage tended to be shorter after Sx-VATS than with 2p-VATS and 3p-VATS (2 d v. 3 d v. 5 d, respectively; p = 0.035). This reflected a shorter, although not significant, overall length of stay of 3 days among patients undergoing Sx-VATS. Finally, although there was no difference in the incidence of minor or major complications between the subgroups, we noted more major complications in the 2p-VATS and 3p-VATS groups (14/97 [14.4%] and 14/87 [16.1%]) than in the Sx-VATS group (1/17 [5.9%]). The type and incidence of all complications stratified by surgical type can be found in Table 4 for Clavien–Dindo grade of 3 or lower, and in Table 5 for Clavien–Dindo grade of 3 or higher.
Perioperative, postoperative, and pathological characteristics according to surgical approach
Type and incidence of all Clavien–Dindo grade ≤ 3 complications stratified by surgical type
Type and incidence of all Clavien–Dindo grade ≥ 3 complications stratified by surgical type
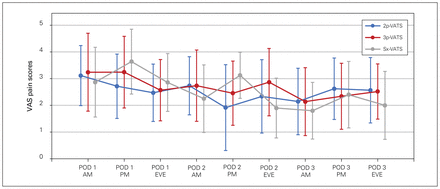
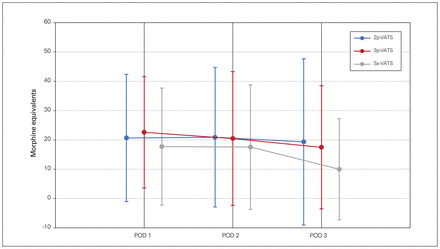
The VAS pain scores for the first 3 postoperative days are shown in Figure 2 and the morphine equivalent scores in Figure 3. We observed no differences among the mean pain scores at each discrete time point for the first 3 postoperative days (p = 0.63). Finally, the morphine equivalents were also similar between the groups.
Quantification of pain using a visual analogue scale (VAS) during the first 3 postoperative days according to surgical approach. 2p-, 3p-, Sx-VATS = 2-port, 3-port, or subxiphoid video-assisted thoracic surgery; AM = morning; EVE = evening; PM = afternoon; POD = postoperative day.
Quantification of pain using morphine equivalences (in milligrams) during the first 3 postoperative days according to surgical approach. 2p-, 3p-, Sx-VATS = 2-port, 3-port, or subxiphoid video-assisted thoracic surgery; POD = postoperative day.
Discussion
In this study, we compared the postoperative outcomes and pain associated with 3 different surgical approaches to thoracoscopic lobar and sublobar resection. Among a group of demographically and clinicopathologically similar patients, we found no statistically significant differences in terms of intraoperative variables, perioperative outcomes, and early postoperative pain scores. However, there was a tendency to a reduced incidence of perioperative complications, shorter pleural drainage and length of hospital stay after the Sx-VATS approach.
Several studies have shown statistically similar intraoperative blood loss and postoperative length of stay when comparing the multiport approaches (2p-VATS and 3p-VATS) with the subxiphoid approach.9,12–15 Conversely, in our study, the biggest difference between the surgical approaches was the shorter length of stay among patients undergoing a Sx-VATS procedure. This is clinically important, as a decreased length of stay is associated with a substantial reduction in the overall cost of a patient’s admission.16 However, some studies have found that the Sx-VATS approach was associated with a longer operative time.13–15 A possible explanation of difference is that the subxiphoid lobectomy is a relatively new approach. As such, the longer operative time may well represent surgeons gaining experience during their learning curve.17
Randomized controlled trials have shown the superiority of VATS over open lobectomy in terms of pain, quality of life, and complication rate.18,19 Thereafter, data comparing complication rates between different VATS approaches have been described. In fact, our analysis is in accordance with the published literature that found no differences in the incidence of postoperative complications when the various VATS approaches were compared.13–15 Nevertheless, in our study, the Sx-VATS group had a trend toward fewer complications, and especially fewer major complications. This may be due to a more risk-averse approach when using a new surgical technique. It is important to note that despite the reduction in the incidence of complications, in the event of a major bleed, achieving hemostasis may be more challenging via the Sx-VATS approach. This could result in a lower threshold for performing an unplanned thoracotomy. Whether the surgical approach affects the incidence of postoperative complications has also been explored in the setting of intercostal robotic lobectomy and sublobar resection. A recent trial comparing robotic-assisted lobectomy with video-assisted lobectomy found that a robotic approach had no impact on the incidence of postoperative complications.20 However, data also suggest that a robotic approach is associated with less postoperative pain, possibly because of reduced torque on the ribs and, in turn, the intercostal nerves.21
With that said, it is thought that fewer intercostal incisions are associated with less postoperative pain.6,8,22 In fact, studies have concluded that uniportal VATS was statistically superior to 3p-VATS regarding postoperative pain in the first 3 days after surgery.22–25 Further data suggest that by avoiding an intercostal incision, such as in the subxiphoid approach, pain can be reduced even more.14,17,26 Despite these findings, our study failed to find any meaningful differences in postoperative pain between the different surgical approaches. This may be explained by the consistent usage of an intercostal block in every patient who underwent surgery, which has been shown to significantly reduce postoperative pain.27
In the cohort presented here, 2 patients with Stage 3 disease underwent Sx-VATS resection. Of note, neither of these patients received neoadjuvant therapy; nor did they have bulky hilar lymphadenopathy on preoperative imaging. As such, in this study we have not assessed the ability to use this approach in the occasionally technically challenging setting of postneoadjuvant therapy, or in those with a challenging mediastinal dissection. Further research is needed to evaluate the oncologic efficacy of this approach in this specific subgroup of patients.
Limitations
Our study is limited by its retrospective nature, and the small sample size of patients included in the Sx-VATS group. The use of the VAS score as the main metric for patient-reported pain is also a limitation. Given the influence of the time during the preceding day that the procedure was performed, the subjective nature of pain and the inability to validate the scores against milligram equivalents of opioids required throughout the day, the generalizability of the results described here is limited. The short follow-up period of only 3 days limits the power of the study as we could not evaluate the incidence of chronic pain and other long-term postoperative complications.
The analysis also does not integrate the oncologic efficacy of each surgical approach in terms of lymph node harvest and potential impact on survival. Future studies should focus on comparing the overall survival of all 3 surgical approaches.
Conclusion
The results of this study suggest that the total number and location of ports used to perform VATS do not significantly affect early perioperative outcomes and postoperative pain scores in patients undergoing ALR for lung cancer. Given the limitations described above, further studies with longer follow-up intervals are required to explore the lasting impact of the surgical approach on quality of life.
Footnotes
Contributors: Jonathan Spicer supervised the study. Jonathan Spicer, Caroline Huynh, and James Tankel contributed to the conception and design of the study. Justin-Pierre Lorange and Amit Katz acquired the data and Justin-Pierre Lorange analyzed it. Justin-Pierre Lorange and Amit Katz wrote the article, which was reviewed by James Tankel, Caroline Huynh, and Jonathan Spicer. All authors approved the final version to be published.
Competing interests: Jonathan Spicer reports receiving grants or contracts from Bristol Myers Squibb, Merck, AstraZeneca, Protalix Biotherapeutics, CLS Therapeutics, and Roche; consulting fees from Bristol Myers Squibb, Roche, Merck, AstraZeneca, Regeneron, Novartis, Eisai, Amgen, and Pfizer; payment or honoraria from Peerview, Onclive, and Medscape; and support for attending meetings or travel from AstraZeneca, Bristol Myers Squibb, and Merck. He also reports participating on advisory or data safety monitoring boards for AstraZeneca, Bristol Myers Squibb, and Merck, and has received equipment, materials, drugs, medical writing, gifts, or other services from Roche, AstraZeneca, Merck, Bristol Myers Squibb, CLS Therapeutics, and Protalix Biotherapeutics. Dr. Spicer is an associate editor of CJS; he was not involved in the review or decision to accept this manuscript for publication. No other competing interests were declared.
- Accepted October 16, 2023.
This is an Open Access article distributed in accordance with the terms of the Creative Commons Attribution (CC BY-NC-ND 4.0) licence, which permits use, distribution and reproduction in any medium, provided that the original publication is properly cited, the use is noncommercial (i.e., research or educational use), and no modifications or adaptations are made. See: https://creativecommons.org/licenses/by-nc-nd/4.0/