Abstract
The development of a cancerous tumour at the site of total joint replacement is a rare but virtually always fatal event. The previously unreported scenario of a patient who was found to have a malignant fibrous histiocytoma at the bone–cement membrane after revision for a loose total hip prosthesis is reported. Recent biologic information evaluating the response of mesenchymal cells to metallic debris suggests that the environment surrounding a loosened prosthesis may provide conditions appropriate for the development of a sarcoma.
About 20 cases of bone sarcoma arising at the site of total joint replacement have been documented in the literature.1–17 All the patients presented with an obvious tumour mass that was readily evident on physical examination or radiographic evaluation. Even when the diagnosis was not suspected before surgery, a sarcoma was obvious on surgical exploration.
We report on a patient who underwent revision arthroplasty for a symptomatic loose prosthesis. No tumour was encountered at surgery, but a sarcoma was identified on histologic examination of the membrane removed from the bone–cement interface. This unusual complication of arthroplasty implantation has not been reported before and raises interesting questions related to the pathogenesis of sarcoma after joint replacement.
Case report
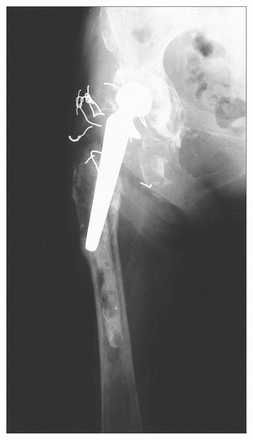
A 62-year-old woman was treated surgically for advanced osteoarthritis of the right hip, resulting from congenital hip dysplasia. A cobalt-chrome femoral prosthesis was implanted with the use of methylmethacrylate cement. Cement was also used to fix a polyethylene acetabular cup. The patient was free of pain for approximately 4 years, when progressive thigh pain began to develop. Seven years after the index arthroplasty she was referred to our hospital for consideration of revision of the prosthesis, which demonstrated radiographic evidence of aseptic loosening and marked proximal osteolysis on the femoral side (Fig. 1).
Before surgery there was extensive resorption of the proximal femur, and the unsupported femoral component had fallen into varus.
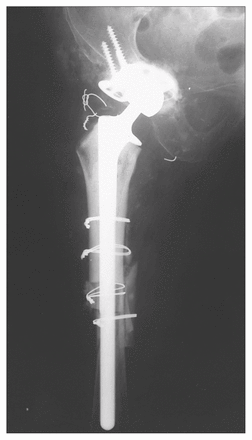
On clinical evaluation the patient gave no history of any malignant disease and reported being well, with the exception of hip pain. Examination of the right hip demonstrated pain on motion and mild tenderness in the previous scar. There was no evidence of a tumour mass on either physical examination or the radiographs. Six months after initial review, the patient underwent revision arthroplasty with a proximal femoral cemented bulk allograft as well as morcellized allograft and a roof ring on the acetabular side. Extensive exposure of the proximal femur and the pelvis was required for re-construction (Fig. 2). The femoral component was extremely loose and was removed manually. Extensive membrane was present at the bone–cement interface of the femur, but no more than would usually be encountered in aseptic loosening of the femoral component. A sample of membrane was submitted for routine histologic evaluation and for culture.
Cemented femoral allograft (press fit distally) was used to reconstruct the proximal femur. A roof reinforcement ring (and morcellized allograft) was used in the acetabulum.
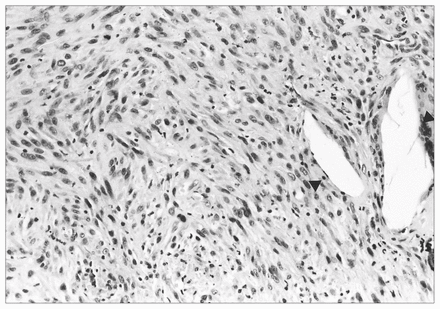
Sections prepared from the membrane demonstrated metallic and polyethylene wear debris together with the usual finding of a histiocytic and giant cell response to foreign material. However, foci of malignant fibrous histiocytoma were also observed infiltrating the inflammatory membrane (Fig. 3).
Histologic appearance of the sarcoma. The cells are spindle shaped, and there is mitotic activity. Giant cell reaction to polyethylene wear debris is present within the tumour (arrowheads) (hematoxylin–eosin, original magnification ×200).
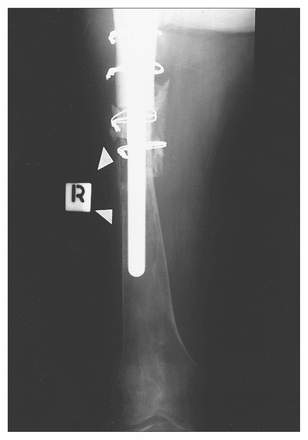
The patient was advised of the finding and elected to undergo no further immediate treatment. Three months after surgery she presented with increasing thigh pain, and two nodules were identified in her wound. Radiographs of the distal femur revealed bone resorption suggestive of tumour infiltration (Fig. 4). Needle biopsies of the nodules in the wound revealed sarcoma, and histologic features identical to the tumour were found in the membrane.
By 4 months after surgery there was extensive endosteal erosion (arrowheads) of the distal femur.
Systemic staging revealed no evidence of metastatic disease, and the patient was counselled to undergo a hindquarter amputation. She refused this treatment. Four months later she died of progressive local and systemic disease. No autopsy was permitted by her family.
Discussion
The patient we describe had microscopic sarcoma arising in the membrane surrounding a loose femoral component, with no gross evidence of a tumour mass. The extensive exposure necessary to reconstruct the proximal femur demonstrated that there was no clinically evident sarcoma that might have extended microscopically to the cement–bone interface. Indeed, there was no clinical sign of the tumour until 3 months after surgery, when nodules were noted in the surgical scar and resorption of the femur distal to the allograft became evident.
This clinical scenario of sarcoma arising in the membrane of a loose total hip prosthesis has not been described before. Of the 20 or more cases of cancer associated with implants reported in the literature, some can be characterized as probable metastatic malignant disease originating at another primary site.13 An additional group of cases are notable for a very short latency period (the time from implantation of the prosthesis to recognition of the cancer).1,6,17 It is possible that these tumours, arising within 2 years of the time of implantation, may have been present at the time of initial surgery and simply were not recognized.
In the remaining cases, a sarcoma arose at the site of an implant 2 to 11 years after the index procedure. 2–5,7–12,14–16 In each case the sarcoma was recognized by the presence of a clinical mass or focal bone destruction on radiographs, a discrete lesion was noted either before or at the time of surgery, and the biopsy of this lesion revealed sarcoma. In no case was the sarcoma recognized only on histologic examination of the bone–cement membrane.
In this case there was no focal lesion. Although bone lysis was identified around the femoral component, it was diffuse and did not progress markedly during the 6 months from our initial encounter with the patient to the time of revision surgery. Therefore we assume that during the time before revision, sarcoma was developing in the bone–cement membrane surrounding the loose femoral component. It is likely that the progression of this lesion would have resulted in the growth of a clinically evident mass had the revision surgery been delayed longer.
Development of sarcoma in the bone–cement membrane has never been reported to our knowledge. In the case that is most comparable to ours, sarcoma was recognized 8 months after a revision procedure, and one would assume that sarcoma was indeed present at the time of revision.16 However, evaluation of the submitted bone–cement membrane tissue from that revision operation failed to demonstrate sarcoma.
The case we report here suggests that sarcoma can indeed arise in the membrane tissue associated with aseptic prosthetic loosening. The pathogenesis of sarcoma in association with metallic wear debris has been evaluated by several investigators, who were able to demonstrate traces of metal in the sarcoma tissue removed from the vicinity of total joint replacements.8,15,18 It is recognized that metal wear debris injected into laboratory animals may result in the development of cancers, 19–23 and it is known that workers exposed to nickel and chromate are at increased risk for epithelial malignant disease.24–26 Recent biologic information evaluating the response of mesenchymal cells to metallic debris suggests that the environment surrounding a loosened prosthesis may provide conditions appropriate for the development of a sarcomatous tumour.
Mahoney and colleagues27 investigated the effect of metallic particulates on cultured synoviocytes in vitro. In addition to measuring proteolytic enzymes and inflammatory mediators produced in response to the material, these investigators also assessed the proliferative effects of particulates (i.e., stimulation of cell division by materials). Titanium, titanium-aluminum and chromium all stimulated DNA formation at low concentrations, whereas cobalt was toxic and nonproliferative even at low concentration. The authors suggest that the proliferative effect of particulates on fibroblasts might account for the fibrous membrane found at the surface of prostheses. Uncontrolled mesenchymal cell proliferation is also present in all sarcomas.
Using both immunohistochemical markers (which detect protein) and in-situ hybridization (demonstrative of mRNA transcripts), Jiranek and associates28 evaluated membranous tissue obtained at revision arthroplasty from 10 loose prostheses. The investigation demonstrated that both interleukin- 1B and platelet-derived growth factor (PDGF) were produced by macrophages and fibroblasts in the periprosthetic membrane. PDGF production is frequently identified in primary sarcoma cultures, and along with insulin-like growth factor, PDGF is one of the most active agents causing mesenchymal cell proliferation.29
If the local environment surrounding a loosened arthroplasty is appropriate to induce mesenchymal cell proliferation and possibly the development of a sarcoma, what risk does this represent and should this risk be taken into account in planning reconstruction of the arthritic hip joint? If one assumes that (a) hip arthroplasty has been used extensively for about 30 years, (b) in those 30 years an average of half a million hip replacements have been performed annually and (c) that each patient has lived with the hip in situ for 20 years, it is possible to calculate the potential risk of a sarcoma developing in the hip that might be entirely coincidental and not attributable to the reconstruction. The risk of a high-grade bone sarcoma developing is 2 cases per million population per year. Most cases of bone sarcoma occur in the first 3 decades of life and tend to be most frequent around the knee, so let us assume that only 0.02 cases of high-grade sarcoma per million patients will present in the hip of individuals older than 50 years.30 If we accept that there are 10 million people in the world with hip replacements (500 000 hip replacements performed annually all lasting 20 years on average) and that this population has been at risk of sarcoma over 30 years, it is simple to calculate that about 6 cases of sarcoma should have developed, by coincidence, with the implant. The fact that only about 20 well-documented cases have been described suggests that the risk of sarcoma is slightly higher in the total population of hip replacements than one might expect by coincidence. This slight increased risk is not a major clinical concern. However, there are three issues that should be considered in evaluating this risk.
The first issue is the underreporting of sarcoma in the total hip replacement population. Most total hip reconstructions are performed by surgeons working outside teaching centres, and these physicians may not report the single case of sarcoma in their joint reconstruction practice. This risk of nonreporting would be especially high, since most patients suffering this complication would be elderly and would likely die rapidly as a result of the sarcoma.
Two further concerns relate to the length of time that patients are living with implants and the type of implants being used in younger patients. Joint replacements are probably being performed for younger patients, who are more likely to be treated by boneingrowth components utilizing a modular head and stem design. Since the metallic debris generated by these implants may exceed the debris observed with cemented components,31,32 it is possible that the risk of sarcoma will increase with increasing service time of uncemented implants.
- Accepted May 24, 1996.